Surfactant
Fundamental Knowledge of Surfactants
Generally, an “interface” refers to the border area between two materials of different states of solid, liquid or gas. “Surfactants,” short for surface active agents, are chemical compounds that exhibit functions and improve the performance of these interfaces.
Basic Structure of Surfactants
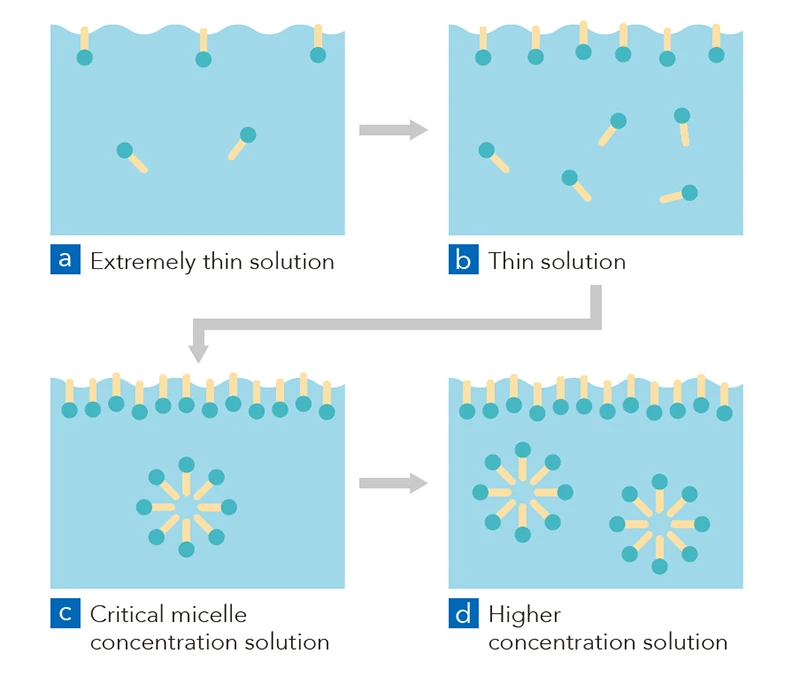
Surfactants have a unique chemical structure that has both hydrophilic (water-attracting) and hydrophobic (oil-attracting) properties. Using this structure, surfactants can achieve a variety of effects such as emulsification, dispersion, foaming, and adsorption by weakening surface tension or forming molecular aggregates or micelles (spheres). When surfactants are dissolved in water and the result is a low-concentration solution, their molecules can easily gather and assemble on the interface (surface) of the solution through a phenomenon called “adsorption.” If the surfactant concentration is increased, the surfactants form molecular aggregates or micelles (spheres) when they reach what is called “critical micelle concentration” (see illustrations “c” and “d” below).
Once micelles have formed, if you add non-water-soluble oil to the solution, the oil can be encircled (solubilized) by the micelles so that from the outside, it looks like the oil has dissolved in the water.
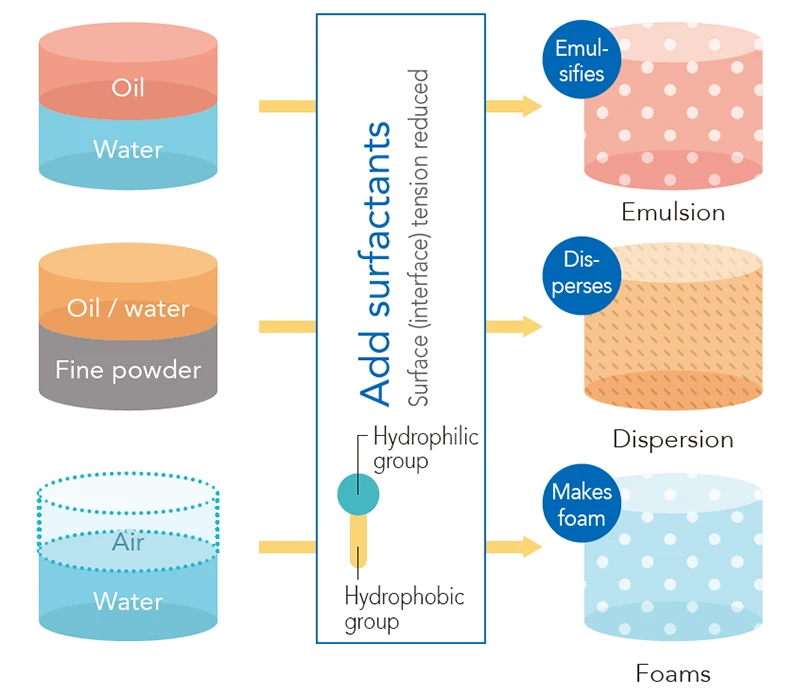
Surfactant Types
Surfactants have four main structural types based on the functions they are molecularly designed to achieve. Of these four types, three are ionic surfactants that transform into electrolytic dissociation ions (atoms or groups of atoms bearing an electrical charge) when dissolved in water, and the remaining type is nonionic surfactants, which do not form ions. The three ionic surfactants are further subdivided based on the type of ion they form when dissolved in water: anionic (or negative ion) surfactants, cationic (or positive ion) surfactants, and amphoteric (containing both positive and negative ions) surfactants.
to scroll
| Types of surfactants | Characteristics | Main applications | Flagship Product Line |
|---|---|---|---|
| Anionic surfactants |
|
Laundry detergent / Shampoo / Body wash | HITENOL series products |
| Cationic surfactants |
|
Hair conditioner / Fabric softener / Sterilant | CATIOGEN series products |
| Amphoteric surfactants |
|
Body wash / Dishwashing liquid / Shampoo | AMOGEN series products |
| Nonionic surfactants |
|
Laundry detergent / Emulsifier and solubilizer / Dispersant / Metal processing oil | NOIGEN series products |
Main Actions and Applications
to scroll
| Function | Actions and effects | Applications | |
|---|---|---|---|
| Emulsification | Mixes incompatible substances | Mixes water and oil to form emulsions. |
Food, cosmetics, paints, dyes  |
| Dispersion | Breaks solid particles into finer pieces and disperses them evenly in a dispersion medium | Mixes inorganic fillers into a dispersion medium to produce nano-dispersions. |
Battery materials, information electronic materials, cosmetics  |
| Moistening, permeating, wetting |
Facilitates substrate wetting and liquid permeation | Enhances the wetting properties of a leaf for uniform, thin pesticide application. |
Agriculture, pesticides, textile paints, dyes  |
| Foaming, defoaming |
Makes and/or removes foam | Uses a foaming agent to introduce air into concrete for molding. |
Civil engineering, construction soaps, detergents, food, cosmetics  |
| Cleaning | Removes dirt | Adsorbs onto dirt, penetrates between the dirt and the substrate, loosens it, breaks it into smaller particles, emulsifies it, and prevents it from reattaching to the substrate. |
Soaps, detergents, electronic component cleaners  |
| Softening, smoothing |
Softens and smooths | Creates soft fabric with gentle feel on the skin. Improves the smoothness of yarns in the spinning and/or knitting process. |
Textile finishing agents, metal processing oils  |
| Antistatic | Prevents static electricity on substrate surfaces | Adsorbs moisture from the air onto substrate surfaces and reduces electrical resistance. |
Electronic substrates, films, resins  |
| Rustproofing | Prevents rust on metal surfaces | Forms a protective film that adheres to metal surfaces to block oxygen and water, which cause rust. |
Metal surface treatment films, electronic wiring  |
| Sterilizing | Removes bacteria | Uses the positive charge of the surfactant to adsorb negatively charged bacteria, disrupting their cell membrane. |
Disinfectants, soaps, detergents  |
Environmental Impact of Surfactants
Most household wastewater that contains surfactants is collected and treated at public sewage treatment facilities; however, some may enter rivers or soils untreated.
Because surfactants are biodegradable, they eventually degrade to carbon dioxide and water by bacteria, even if released into the environment. To preserve the natural environment, products with high biodegradability are being developed and proactively used in Japan.











