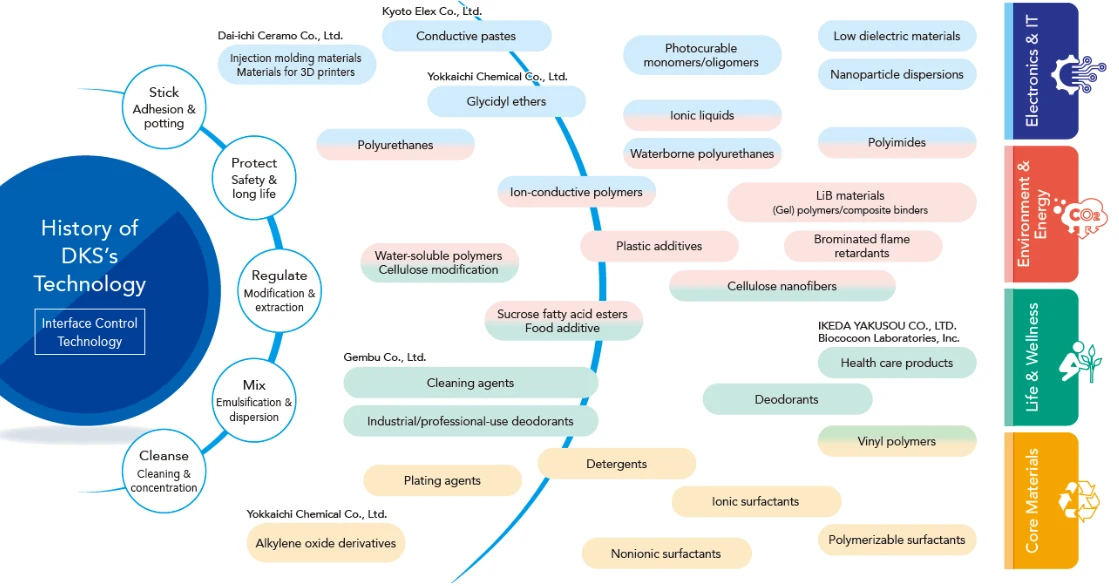
Strengths of DKS Technology
Our strength lies in our technological capability to optimally customize materials to the functions and performance our customers require, and to propose the best solutions by combining multiple technologies. Centered on “the interfacial control technologies” we have refined since our founding, we integrate a wide range of technologies to supply high-performance materials to diverse industrial fields.
In recent years, we have focused on developing materials that help address social challenges such as digitization, achieving a decarbonized society, improving health and well-being, and promoting resource circulation.
to scroll
Fundamental Technologies
DKS has four business segments, in line with the DKS Credo of “Contributing to the nation and society through industry.” DKS boasts an optimal combination of technological strengths that address the function and manufacturing method-related needs of each customer through customized proposals. DKS has cross-disciplinary fundamental technologies cultivated throughout our history since 1909. By combining these technologies, adding new developments, and continuously innovating, we will keep realizing “Chemistry Provides a Solution.”
Surfactants
Surfactants are substances that possess both hydrophobic and hydrophilic properties and modify the characteristics of interfaces between immiscible substances.
For example, oil and water normally separate into two layers; however, the addition of a surfactant enables emulsification, allowing two immiscible liquids to form a homogeneous mixture. When insoluble solid particles (powder) are added to a liquid, aggregation and sedimentation tend to occur, but adding a surfactant makes it possible to disperse the particles homogeneously. Surfactants also form foam at the interface between air and water.
When surfactants are added to water at concentrations above a certain level, micelles (aggregates) are formed. Micelles are colloidal particles in which molecules assemble with their hydrophobic groups oriented inward and hydrophilic groups outward. These structures can encapsulate various poorly water soluble substances and disperse them in water.
By bridging dissimilar materials, surfactants contribute to the realization of diverse performance requirements demanded by customers and society.
Sucrose or Cellulose Derivatives
Derivatives are compounds in which part of a molecule is substituted with another atom or functional group through chemical reactions while maintaining the basic molecular framework.
Sugars are a class of carbohydrates represented by the general formula Cm(H₂O)n. In a narrow sense, the term refers to water soluble monosaccharides and disaccharides. A representative example is sucrose, the main component of table sugar. Sucrose is a disaccharide composed of α glucose and β fructose.
We offers a range of sucrose derivative products, including sucrose fatty acid esters (SE), sucrose benzoates (SB), and sucrose octaacetates (SOA).
Polysaccharides produced by plant photosynthesis include starch, which humans can digest, and cellulose, which cannot be digested. Cellulose consists of linearly linked β glucose units. Because the molecular chains are aligned in parallel and stabilized by hydrogen bonding, cellulose is poorly soluble in water. However, by converting the hydroxyl groups (–OH) on the glucose units into various functional groups, derivatives such as sodium carboxymethyl cellulose (CMC) and cellulose nanofibers (CNF) can be synthesized.
These sugar and cellulose derivatives are biomass based materials derived primarily from renewable plant resources and are attracting increasing attention as key materials for realizing a sustainable society.
Sucrose Fatty Acid Eters (SE, Sugar esters)
Sucrose is a disaccharide composed of α glucose and β fructose and is the main component of sugar, characterized by its hydrophilic nature. Fatty acids, on the other hand, are hydrophobic compounds consisting of a hydrocarbon chain with a single terminal carboxyl group (–COOH). Sucrose fatty acid esters (SE) are produced by chemically bonding these components through esterification.
Sucrose fatty acid esters are classified as sugar derivatives and are nonionic surfactants that exhibit both hydrophilic and hydrophobic properties. Their high level of safety has been recognized by international organizations, including the FAO/WHO Joint Expert Committee on Food Additives, and they were approved as food additives in Japan in 1959.
Owing to their excellent emulsifying performance, ability to solubilize poorly soluble substances, foaming properties, and high safety, sucrose fatty acid esters are widely used in food applications such as processed foods, confectionery, and dairy products, as well as in pharmaceutical and personal care applications worldwide.
Sodium Carboxymethyl Celluloses (CMC)
Sodium carboxymethyl cellulose (CMC) is a water soluble anionic polymer obtained by introducing carboxymethyl groups into the cellulose backbone. It combines excellent thickening and stabilizing performance with low environmental impact due to its biodegradability.
As a result, CMC is widely used as an additive in papermaking and animal feed applications.
In aqueous solutions, hydrophobic interactions along the cellulose main chains provide strong adsorption onto hydrophobic particles, exhibiting protective colloid behavior. Leveraging this property, CMC has also been adopted in recent years as a dispersion stabilizer for active materials in lithium ion batteries (LIBs).
Cellulose Nanofibers (CNF)
Cellulose is a naturally occurring polymer derived from plants. Cellulose nanofibers (CNF) are produced by fibrillating cellulose fibers contained in wood and other biomass down to the nanometer scale, resulting in materials that are lightweight, high strength, and exhibit low thermal expansion.
CNF is manufactured using a TEMPO mediated oxidation process, which selectively converts hydroxyl groups into carboxyl groups. This method yields CNF with an average fiber width of approximately 3 nm and a length of about 800 nm, providing an extremely high aspect ratio.
As a plant derived and sustainable next generation material, CNF is being applied across a wide range of industrial fields, particularly as a thickener and rheology modifier.
Polyvinylpyrrolidone (PVP)
Polyvinylpyrrolidone (PVP) is a water soluble polymer developed in the late 1930s and is known for its excellent dispersibility, protective colloid properties, film forming ability, and adhesion. In addition to water, PVP dissolves in many polar solvents, including alcohols, acetic acid, chloroform, and amines.
Due to its high level of safety for both humans and the environment, PVP is widely used in medical applications such as disinfectant formulations, tablet binders, ophthalmic solutions, and hollow fibers. In the cosmetics field, it is used in hair styling products and mascaras, while in industrial applications it is employed for semiconductor wafer surface planarization, photoresist formulations, glue sticks, and inkjet printing materials.
Polyurethane
Polyurethane resins are synthetic polymers characterized by urethane linkages (–NHCOO–) in their molecular backbone. By selecting appropriate combinations of polyisocyanates and polyols, a wide variety of structures and physical properties can be designed.
Polyurethanes include elastic materials such as polyurethane rubbers and elastomers, as well as flexible and rigid polyurethane foams produced through foaming reactions. Flexible polyurethane foams exhibit excellent resilience and vibration damping properties and are widely used as cushioning materials for furniture and automobiles. Rigid polyurethane foams have a closed cell structure and superior thermal insulation performance, making them suitable for insulation applications.
Polyurethane prepolymers react with water to cure and are therefore used in waterproofing materials, water stopping agents, and ground consolidation agents for tunnel excavation.
Waterborne Polyurethane Dispersions (Water-Dispersed Polyurethanes, PUDs)
As efforts to reduce environmental impact continue to expand, lowering VOC (volatile organic compound) emissions has become an especially important issue. In this context, waterborne polyurethane resins, also known as waterborne polyurethane dispersions (PUDs), have attracted significant attention.
Polyurethane resins are inherently insoluble in water; however, they can be dispersed in water through self emulsifying systems that impart hydrophilicity or through forced emulsification using surfactants.
PUDs are classified into non reactive types, which are used as is, and reactive types, which undergo curing upon heat treatment. Non reactive types are used in coatings, adhesives, paints, binders, and films, imparting properties such as adhesion, heat resistance, water resistance, chemical resistance, and high hardness.
Reactive types consist of polyurethane prepolymers with blocked isocyanate groups. Upon heating, the blocking agents dissociate, regenerating active isocyanate groups that react and crosslink with compounds containing active hydrogen, thereby providing enhanced modification effects.
UV / EB Curing Resins
UV / EB curing resins are materials that cure when monomers or oligomers are exposed to ultraviolet (UV) light or electron beams (EB). This curing technology, also known as radiation curing, enables instantaneous curing compared with conventional thermal curing methods that require heat, thereby contributing significantly to improved productivity and reduced energy consumption.
In particular, the use of multifunctional monomers allows the formation of cured films with excellent mechanical properties, and as a result, these materials are widely used across various fields. In addition, precisely controlling the addition of alkylene oxides (AO), a technology derived from surfactant chemistry, it is possible to adjust the balance between hydrophilicity and hydrophobicity of the monomers, enabling lower viscosity and faster curing speeds.
UV curable oligomers, such as urethane acrylates and epoxy acrylates, can impart a wide range of properties—from hardness to flexibility, as well as high refractive index, low curl, strong adhesion, and impact absorption—to coatings and films.
Flame Retardants
Flame retardants are widely used to impart flame resistance to plastics, rubber, wood, textiles, and other materials, and they play an important role in reducing human injury and economic loss caused by fires.
Flame retardancy is achieved by interrupting the combustion process. Halogen based flame retardants, such as brominated compounds, suppress the thermal decomposition of polymers that leads to the generation of flammable gases by capturing active radicals and reducing their reactivity. In addition, their decomposition products absorb heat and generate heavier gases, further inhibiting combustion.
In contrast, phosphorus based flame retardants form a molten protective layer and promote char formation by altering thermal decomposition pathways.
Halogen based flame retardants, in particular, offer high flame retardant efficiency and are widely used in plastic applications.
Ionic Liquids
Ionic liquids are compounds composed of cations and anions that exist in a liquid state at temperatures typically below 100°C. Because the Coulombic interactions between ions are relatively weak and do not allow the formation of an ordered crystalline structure, ionic liquids exhibit low melting points and remain liquid.
Often referred to as the “third class of liquids,” following water and organic solvents, ionic liquids offer a wide liquid state temperature range, non flammability, negligible vapor pressure, high thermal stability, high ionic conductivity without added electrolytes, excellent electrochemical properties, and high design flexibility.
These unique characteristics make ionic liquids promising candidates for applications such as high performance antistatic agents, electrolytes for secondary batteries, lubricants, and reaction or extraction solvents.
Deodorizers
The human sense of smell is extremely sensitive and can easily detect even trace amounts of malodorous compounds. At the same time, odor causing substances are highly diverse, making it difficult to identify every contributing component at the source. Moreover, completely removing these substances from an enclosed space is often impractical.
For this reason, the odor neutralization method, which reduces perceived odor intensity and discomfort by combining malodorous components with other fragrances—even when the odor itself is still present—is recognized as an effective approach to odor control.
Our deodorant products are developed by certified Olfactory Measurement Operator, a nationally recognized qualification in Japan, by applying the principles of odor neutralization.
Please refer to the R&D topics for the latest R&D developments.
Discover! DKS










